Environmental Monitoring Solutions for Pharma, Biotech, Life Sciences, Healthcare and Logistics
For 40 years, ELPRO has been a trusted partner in compliant environmental monitoring. We’re committed to supporting your entire pharmaceutical supply chain—from production and storage to transport and delivery to the end user. With in-house developed hardware and software, GxP-compliant consulting, and global support services, we ensure data integrity, compliance, and peace of mind—every step of the way.
Celebrating 40 Years of Monitoring Excellence
This year, ELPRO marks four decades of pioneering solutions in environmental monitoring. Explore key milestones, innovations, and the journey that shaped 40 years of success.

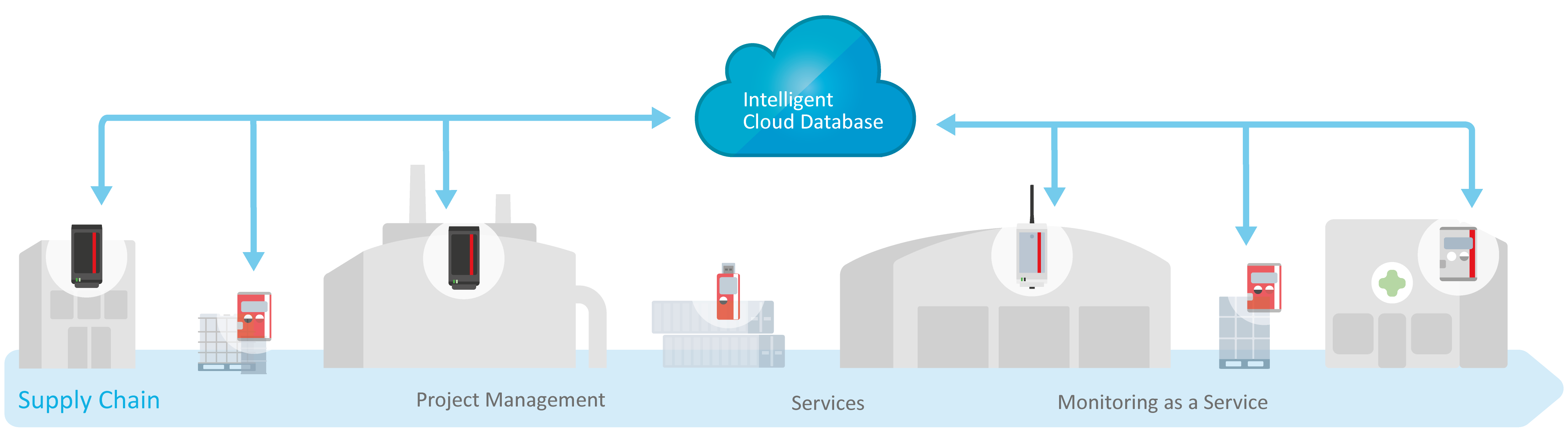
Cold Chain
Solutions for monitoring and analyzing product stability data across the entire supply chain—fully automated.

Room & Equipment
Compliant, scalable solutions for monitoring production areas, storage facilities and equipment.

Consulting, Services & Support
Monitoring as a Service, project management, GxP consulting, qualification, and mappings, and on-/off-site service.
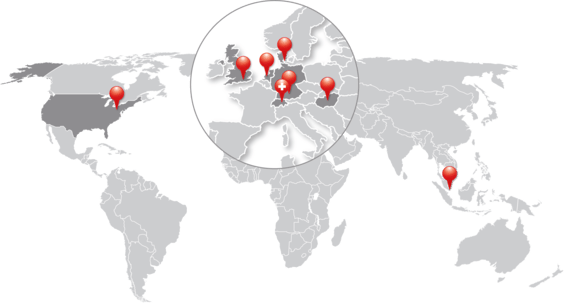
Trusted Experts in Environmental Monitoring Worldwide
Since 1986, ELPRO has supported the pharma, biotech, and healthcare industries with intelligent monitoring solutions—ensuring compliance, visibility, and safety from production to patient. Discover why ELPRO is the right partner to safeguard your supply chain.
40 Years of Excellence
Through its long history, ELPRO has been a trusted leader in environmental monitoring, setting important benchmarks and shaping industry standards worldwide.
8 Locations
ELPRO combines development, production, calibration labs, sales, and service across multiple global sites—ensuring fast, reliable support close to the customer.
4 Calibration Labs
ELPRO operates accredited calibration labs at key locations, making it easy to recalibrate your devices locally and stay compliant with regulatory requirements.
Sustainable Thinking Every Step of the Way
Sustainability is more than a goal—it’s built into everything we do at ELPRO. From product design to end-of-life and circular economy practices, we take responsibility across the entire value chain. Learn how sustainability drives our decisions and shapes the way we do business.

News, Stories and Events
Cold Chain Management: Ensuring Product Quality Across the Supply Chain
IT Teams Must Stay Informed About Temperature and Condition Monitoring Systems (Updated 2026)
Beyond Monitoring: Optimizing Pharma Cold Chain Shipments With Smarter Data Use
From Lab to Launch:
De-Risking the Biologic Cold Chain
June 9-10, San Diego
Powered by ELPRO and Cold Chain Technologies
Complimentary (no fee) to pharma, biotech, life science and logistics leaders in a relaxed, retreat-style setting to network, collaborate and benchmark ideas and challenges. Make plans today to join us. Seating is limited.
Where Leading Pharma Minds Meet
The Leading Minds Network is an engaged peer-to-peer group of pharma, life science, and biotech industry experts and partners sharing unique insights and best practices, so those experiences can be shared for the greater good.
- Peer-to-peer collaboration
- Global onsite seminar series
- “Temperature Control Tuesdays” webinar series
- Member updates & news
- Leading industry experts & partners
- Free to join—no membership fee

Stay up to Date with News and Seminars
Newsletter-Sign Up ELPRO News
ELPRO will use the information provided in this form to keep in touch with you and to send you updates and marketing information by e-mail.